
Newsroom
In a recent study published on Science Advances, researchers led by Prof. LI Xinjian from the Institute of Biophysics of the Chinese Academy of Sciences have uncovered a novel immunoregulatory function of isocyanic acid (ICA), a reactive metabolite synthesized by the enzyme laccase domain containing 1 (LACC1).
The researchers found that ICA plays a crucial role in limiting inflammation by disrupting NLRP3 inflammasome activation in macrophages. The discovery provides new insights into the metabolic regulation of inflammatory responses and potential therapeutic targets for inflammatory diseases.
LACC1, a metabolic enzyme highly expressed in inflammatory macrophages, has long been recognized for its role in breaking down citrulline into ornithine and ICA. While previous studies have associated ICA-induced protein carbamoylation with various diseases, its role in immune regulation remained unclear.
The study demonstrates that lipopolysaccharide (LPS) stimulation triggers LACC1-dependent ICA production, which subsequently modifies NLRP3 and impairs inflammasome formation. at lysine-593, disrupting its interaction with NEK7, a critical step in inflammasome activation. The inhibition of NLRP3 activation ultimately results in an attenuated inflammatory response.
This post-translational modification disrupts the formation of the inflammasome complex, thereby reducing inflammation. Loss of ICA production, either through LACC1 knockout or mutation of lysine-593 in NLRP3, led to an exaggerated inflammatory response in both in vitro and in vivo models.
The findings suggest that ICA acts as a natural inhibitor of NLRP3 inflammasome activation. This discovery opens new avenues for therapeutic interventions targeting inflammasome-driven diseases. Modulating ICA levels or mimicking its effects could provide novel strategies for treating inflammatory conditions linked to NLRP3 dysregulation.
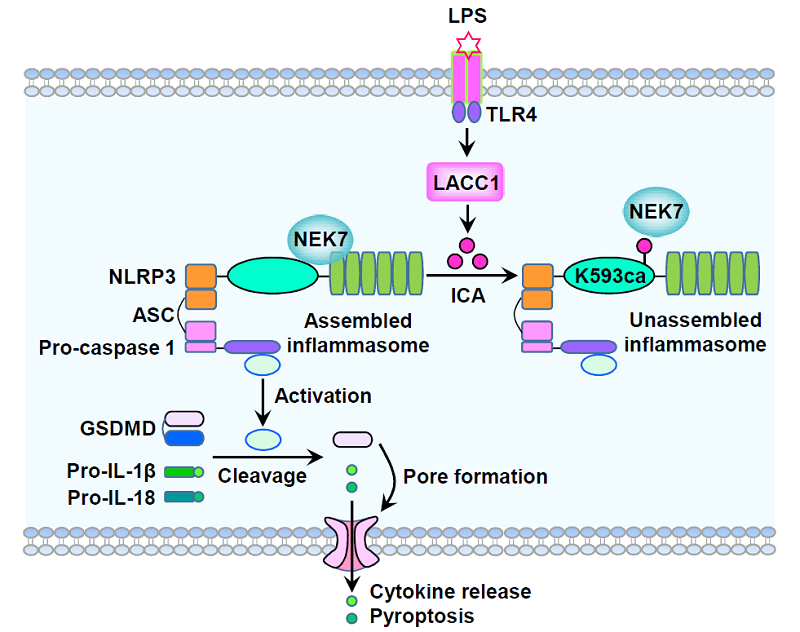
A schematic diagram shows ICA-mediated NLRP3 K593ca limits NLRP3-driven inflammation. ICA, a metabolite synthesized by LACC1 in LPS-stimulated macrophages, directly carbamoylates NLRP3 at K593. ICA-mediated NLRP3 K593 carbamoylation (K593ca) suppresses NLRP3 inflammasome assembly by disrupting the interaction between NLRP3 and NEK7, thereby limiting pyroptosis triggered by NLRP3 inflammasome activation. (Image by LI Xinjian's group)
