
Recently, Prof. TANG Yongbing and his team members (Dr. LANG Jihui, Dr. JIANG Chunlei and FANG Yue) from Functional Thin Film Research Centre, Shenzhen Institutes of Advanced Technology (SIAT) of the Chinese Academy of Sciences, have developed a room-temperature researchable Calcium-ion based hybrid battery with high rate capability and long-term cycling life.
A tri-ion (Ca2+/Li+/PF6-) strategy has been proposed innovatively, leading to high capacity retention of 97% at a current rate of 15 C. Moreover, long cycling life over 1500 cycles at 5 C was also achieved with a capacity retention of 86%.
The research entitled "Room-Temperature Rechargeable Ca-Ion Based Hybrid Batteries with High Rate Capability and Long-Term Cycling Life" has been published in Advanced Energy Materials.
Calcium-ion batteries show great promise as novel energy storage devices benefiting from the high natural abundance, relatively low reduction potential close to lithium and bivalent feature of calcium. However, the development of calcium-ion batteries has been hindered by the irreversible plating/stripping of calcium anode at room temperature as well as the low rate and cycling performance resulted from the large ionic radius and the multivalent nature of calcium ions.
Based on the aforementioned considerations, the team developed a novel calcium-ion based hybrid battery using a tri-ion (Ca2+/Li+/PF6-) strategy.
In this configuration, Sn was selected as the anode which can alloy with both Ca2+ and Li+, while expanded graphite was used as the cathode capable of being intercalated/deintercalated reversibly by PF6- anions.
On the one hand, high reversible intercalation/deintercalation of PF6- into/from the graphite cathode replaces the sluggish kinetics of calcium ions in the conventional cathode materials, which results in low rate performance and cycling stability. On the other hand, the reaction kinetics with the Sn anode can be enhanced significantly by hybridizing a small amount of lithium ions with smaller ionic radius and faster diffusion kinetics.
The research results showed a high rate capability up to 15 C with a capacity retention of 97% and excellent cycling stability over 1500 cycles at 5 C with a capacity retention of 86, which are the best results of reported calcium-ion based full batteries.
This work has provided a novel approach to improve the electrochemical performance of calcium ion batteries at room temperature.
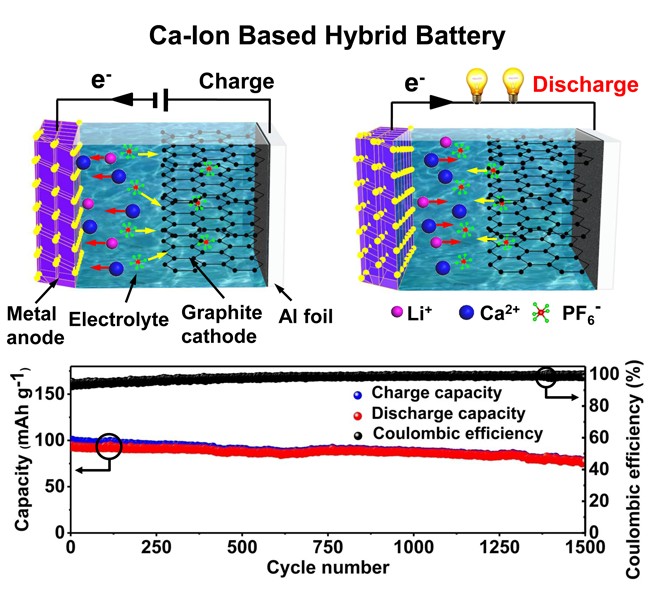
Fig. Diagrammatic drawing of the calcium-ion based hybrid battery (upper) and the long-term cycling performance (below) (Image by Prof. TANG)

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

