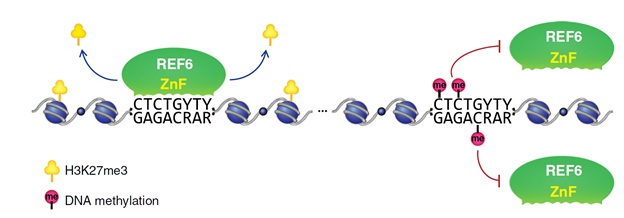
Polycomb-mediated trimethylation of histone H3 lysine 27 (H3K27me3), a conserved epigenetic mark associated with chromatin compaction and gene repression, plays a key role in cell identity and developmental regulation in both plants and metazoans. Dynamic regulation of H3K27me3 at specific targets is achieved by both the histone methyltransferase, PRC2 complex, and the JmjC-domain containing demethylases of H3K27me3. Arabidopsis histone demethylase REF6 specifically demethylates H3K27me3 at thousands of genes by directly recognizing the CTCTGYTY motif via its zinc-finger (ZnF) domains. REF6 tends to bind CTCTGYTY-motifs located in active chromatin states and depleted in heterochromatic regions. However, the underlying mechanism remains unknown.
Recently, a team led by Prof. CAO Xiaofeng at Institute of Genetics and Developmental Biology of the Chinese Academy of Sciences (CAS), in collaboration with Prof. MA Jinbiao's team from Fudan University, revealed H3K27me3 demethylase REF6 preferentially binds to hypo-methylated CTCTGYTY-motifs via its zinc-finger domains, suggesting that DNA methylation might deter REF6 binding in heterochromatin.
The researchers found that REF6 prefers to bind hypo-methylated genomic regions in vivo, and that CHG methylation in CTCTGYTY-motif decreases REF6 DNA binding affinity in vitro.
In addition, crystal structure of REF6 ZnF-clusters in complex with DNA oligonucleotides reveal that 5-methylcytosine is unfavorable for REF6 binding.
To further study the effect of DNA methylation on REF6-binding, the researchers profiled REF6-binding sites in drm1 drm2 cmt2 cmt3 (ddcc) quadruple mutants, in which non-CG methylation is significantly reduced. Interestingly, REF6 ectopically bind to a small number of new target loci, most of which are located in or neighbored with short TEs in euchromatic regions.
These results reveal a mechanism that a histone modifying enzyme avoids heterochromatic binding through its intrinsic DNA methylation unfavorable binding activity. It will be interesting to further explore how recruitment of a chromatin-modifying enzyme is tightly regulated to achieve the appropriate level of chromatin modification and maintain proper chromatin status at the right place in the genome.
This study was supported by grants from the National Natural Science Foundation of China, Chinese Academy of Sciences, the Ministry of Science and Technology of China, the Youth Innovation Promotion Association of CAS, and the State Key Laboratory of Plant Genomics.
Model of how DNA methylation prevents REF6 binding to the CTCTGYTY motif (Image by IGDB)