
Oncogenic fusion protein AML1-ETO, generated by the t(8;21) chromosomal translocation, plays an important role in the pathogenesis of t(8;21) leukemia. AML1-ETO exerts its function in a stable protein complex named AML1-ETO-containing transcription factor complex (AETFC).
The members of AETFC complex, HEB and E2A, belong to E protein family, and are both vital to AML1-ETO induced leukemogenesis. Interestingly, E2-2, the third member of E protein family, was found to be absent in t(8;21) leukemia cells, suggesting that it could be a negative regulator of t(8;21) leukemogenesis.
In a recent study published online in PNAS, researchers from Shanghai Institute of Nutrition and Health of Chinese Academy of Sciences, Shanghai Jiao Tong University School of Medicine and Rockefeller University, found that E2-2 abrogates the function of AETFC complex and negatively regulates leukemogenesis. The underlying mechanism involves E2-2–mediated redistribution of AETFC complex to specific target genes, inducing a dendritic differentiation of the leukemic cells.
Scientisits revealed that the overexpression of E2-2 selectively inhibited the proliferation of AML1-ETO–positive leukemic cells, which requires the basis helix-loop-helix (bHLH) DNA-binding domain.
Through RNA-seq and ChIP-seq analyses, they found that the three E proteins differentially regulate many target genes, and in particular E2-2 redistributes AETFC to, and activates, some genes associated with dendritic cell differentiation. In acute myeloid leukemia (AML) patients, the expression of E2-2 is relatively lower in the AML1-ETO–positive subtype, and an E2-2 target gene, THPO, is identified as a potential predictor of relapse.
Moreover, scientists found that inhibition of E2-2 accelerated leukemogenesis in a mouse model of human t(8;21) leukemia.
Taken together, the three E proteins defined a heterogeneity of AETFC complex. This study provided a promising potential for an efficient dendritic differentiation of t(8;21) leukemia and for the development of the immunotherapy for AML1-ETO-associated leukemia.
The study was funded by the National Key Research and Development Plan of China, National Natural Science Foundation of China, Chinese Academy of Sciences, etc.
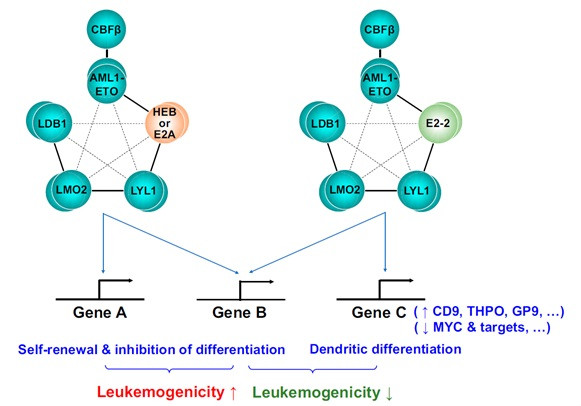
A working model for the different roles of E proteins in AML1-ETO–associated leukemia. (Image by Dr. WANG Lan's group)

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

