
FOXP3+ Treg cells play a central role in maintaining the balance between immune tolerance and activation. Treg cells have a great plasticity and are not stable under inflammation and other physiological conditions. For instance, upon IL-6 stimulationTreg cells can lose the expression of FOXP3 and the suppressive function. However, the molecular mechanism of Treg cell instability under IL-6 stimuli remains largely unclear.
Recently, Dr. LI Dan and KONG Chao supervised by Professor LI Bin at Institut Pasteur of Shanghai (IPS), CAS and Professor SHI Guochao at Ruijin Hospital, Shanghai Jiao Tong University revealed a new molecular pathway by which miR-125a-5p reduces the sensitivity of Treg cells toward IL-6 conversion.
Researchers observed that the expression of miR-125a-5p is lower in resting Treg cells and could be upregulated in a GATA3 dependent manner after TCR activation. IL-6R and STAT3 are the targets of miR-125a-5p in Treg cells. Overexpression of miR-125a-5p increases the stability of Treg cells under IL-6 stimulation and knockdown of miR-125a-5p decreases its stability. TheGATA3/ miR-125a-5p/IL-6R&STAT3/FOXP3 axis is deregulated in Treg cells from PBMC of asthma patients. This research proposes a novel signal pathway in which miR-125a-5p decreases the sensitivity of Treg cellstoward IL-6-mediatedconversion.
The study entitled “MiR-125a-5p Decreases the Sensitivity of Treg cells Toward IL-6-Mediated Conversion by Inhibiting IL-6R and STAT3 Expression” was published online in Scientific Reports on October 1 2015.
This work was supported by grants from National Basic Research Program of China (973 Program), and the National Natural Science Foundation of China.
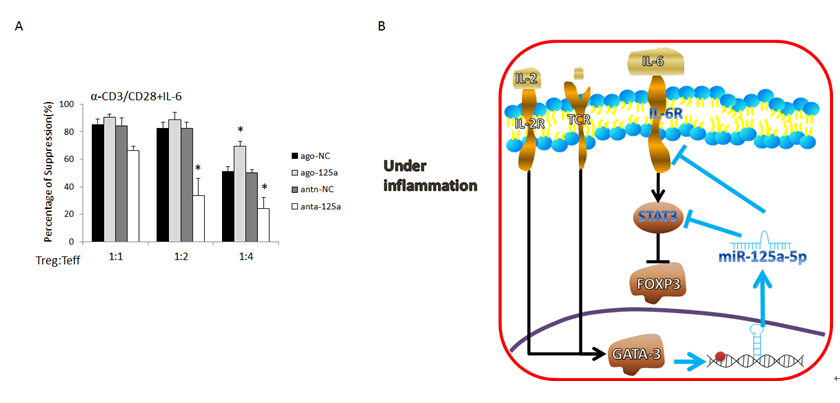
Figure A. The suppressive function of Treg cells under Il-6 stimulation. Figure B. Proposed working model for the mechanism of miR-125a-5pregulatingthe stability of Tregcells. (Image by IPS)

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

