Research group lead by Prof. WANG Wei from SIMM (Shanghai Institute of Materia Medica) collaborating with Prof. LI Jian from East China University of Science and Technology recently made a significant progress on organocatlysis. The work, entitled “Organocatalytic enantioselective beta-functionalization of aldehydes via oxidation of enamines and their application in cascade reactions” has been published by Nature Communications.
Aminocatalysis has become the landmark of organocatalysis. Among them, iminium catalysis involving transformation of iminium ions to enamines has been extensively explored for a number of organic processes. However, the reverse order of an enamine to an iminium species has not been realized.
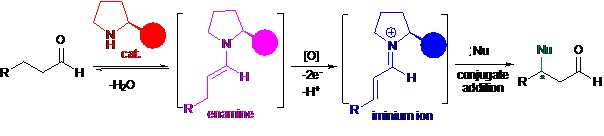
In this work researchers reported a new transformation - oxidative enamine catalysis: direct oxidation of an enamine to an iminium species. Studies reveal that the use of IBX as an oxidant in the presence of a secondary amine catalyst serves as effective system for rapid conversion of enamines to iminium ions. The power of the chemistry has been demonstrated for the direct asymmetric beta-functionalizations of simple aldehydes.
Moreover, a variety of novel enantioselective cascade transformations including triple and quadruple cascades have been realized for the “one-pot” preparation of versatile chiral building blocks and frameworks from simple aldehydes. The operational simplicity and mild reaction conditions render the chemistry particularly attractive in the practice of organic synthesis.
Paper abstract:
http://www.nature.com/ncomms/journal/v2/n3/full/ncomms1214.html
The mechanism of oxidative-dehydrogenation-addition cascade reactions (Image by SIMM)