
Cytotoxic T lymphocytes (CTL) and natural killer (NK) cells defend the body against virus-infected and tumor cells. A key step in this defense mechanism is the release of cytotoxic granules which contain the pore-forming protein perforin and different granule-associated proteases, including granzymes. A variety of granzymes, which are structurally related serine proteases that differ in their substrate specificity, have been described. While granzyme A and B have been extensively studied, relatively little is known about granzyme F (GzmF).
Research on NK/CTL-mediated cell defense mechanisms against cancers and viruses is one of the key areas of research in Prof FAN Zusen's lab. In their recent Cell Death and Differentiation paper1 they report how they discovered that GzmF causes a novel cell death pathway that is distinct from typical apoptosis and then describe how they characterized this pathway.
Murine GzmF is highly expressed in NK3.1 cells and in lymphokine-activated killer (LAK) cells. By introducing GzmF into tumor cells and examining its effects, Prof Fan's group discovered that GzmF causes dramatic cytolysis, thus inducing cell death. It also causes externalization of phosphatidylserine, nuclear condensation, mitochondrial damage, cytochrome c (cyt c) release, caspase inactivation and single-stranded DNA nicking. They observed that GzmF-induced chromatin is incompletely condensed and is segmented at the nuclear periphery. Cellular organelles were damaged and the cytoplasm showed extensive vacuolization that is reminiscent of necroptosis. GzmF causes rapid mitochondrial swelling, depolarization and reactive oxygen species accumulation. Prof Fan's group showed that GzmF-induced death does not involve caspase activation, Bid cleavage or activation of the DNA nickase NM23H1. GzmF-silenced LAK cells showed reduced cytotoxicity against caspase-inhibited tumor cells, and cyt c release was independent of the pro-apoptotic proteins Bid and Bax/Bak. GzmF acts by impairing mitochondrial electron transport and thus abolishes ATP generation. ATP decline may contribute to the failure of apoptosomes to form, leading to caspase inactivation.
This research was funded by the National Natural Science Foundation (Grant No. 30525005, 30830030, 30772496, 30700126), the 863 program (Grant No. 2006AA02Z4C9), the 973 program (Grant No. 2006CB504303, 2006CB910901), the Innovation program (Grant No. KSCX2-YW-R-42) and the 100 Talents Program of the Chinese Academy of Sciences.
1 L Shi, L Wu, S Wang and Z Fan (2009) Granzyme F induces a novel death pathway characterized by Bid-independent cytochrome c release without caspase activation. Cell Death and Differentiation 16, 1694–1706. doi:10.1038/cdd.2009.101
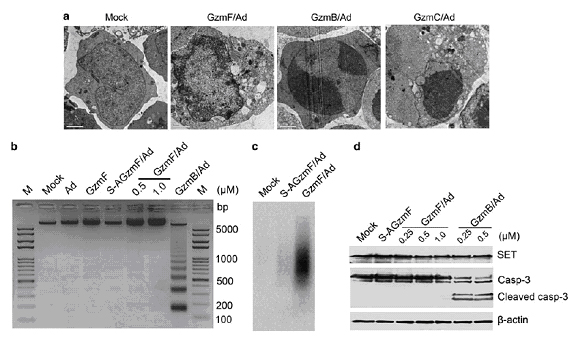
Figure GzmF causes nuclear morphological changes with single stranded DNA nicking. (a) Nuclear morphological changes induced by GzmF. Yac-1 cells were treated by 0.5 μM GzmF, GzmB or GzmC in the presence of Ad at 37°C for 6h. Cells were harvested and visualized by transmission electron microscopy (magnification × 1700). GzmB/Ad and GzmC/Ad were used as contrast. (b) GzmF does not cause DNA laddering. Yac-1 cells were treated with S-AGzmF (1 μM) and the indicated concentrations of GzmF plus Ad at 37°C for 6 h. The genomic DNA was extracted and separated on 2% agarose gel stained with ethidium bromide. M, DNA marker. (c) GzmF induces single stranded DNA nicks. GzmF/Ad treated Yac-1 cells were radiolabeled with 32P-dATP by Klenow fragment. Then the genomic DNA was harvested and visualized by denaturing alkaline gel electrophoresis. These data are representative of at least three independent experiments. (d) GzmF does not cleave SET. Yac-1 cells (3×105) were treated with different concentrations of GzmF or GzmB plus Ad. Samples were harvested and detected with anti-SET antibody. Caspase-3 (casp-3) was probed as a positive control for GzmB. β-actin was used as a loading control.

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

