
A research team led by Prof. HUANG Qing from the Hefei Institutes of Physical Science of the Chinese Academy of Sciences proposed a new nanocomposite based on Bi2MoO6/MoS2/AuNRs for near-infrared (NIR)-II light-boosted photodynamic/chemodynamic therapy.
The study was published in Langmuir and recommended as the journal's cover.
Bi2MoO6 (BMO) nanoparticles have been widely used in photocatalytic applications. They have also been used as photosensitizers in photodynamic therapy. However, their UV absorption property hinders the clinical application.
In this study, the researchers designed a new nanocomposite named Bi2MoO6/MoS2/AuNRs (BMO-MSA) by modifying Bi2MoO6 (BMO) by MoS2/AuNRs (MSA) hybrid. They found that the resulting nanocomposite can absorb light in the NIR-II range.
When exposed to light at a wavelength of 1,064 nm, BMO-MSA produced singlet oxygen (1O2) with a quantum yield of 0.32, confirming its photodynamic therapy (PDT) ability. Moreover, it's peroxidase-like activity enhanced the effect of chemodynamic therapy (CDT).
To investigate the in vivo PDT efficiency of BMO-MSA, the researchers examined the germline apoptosis based on their previously established C. elegans PDT model.
The results showed that PDT induced germline apoptosis in the worm through the cep-1 pathway due to DNA damage, which were further supported by the use of a variety of mutants that lack the function of DNA damage genes.
This work not only provides a novel PDT agent that can be used for PDT in the NIR-II region, but also introduced a new approach to therapy that exploits both PDT and CDT effects.
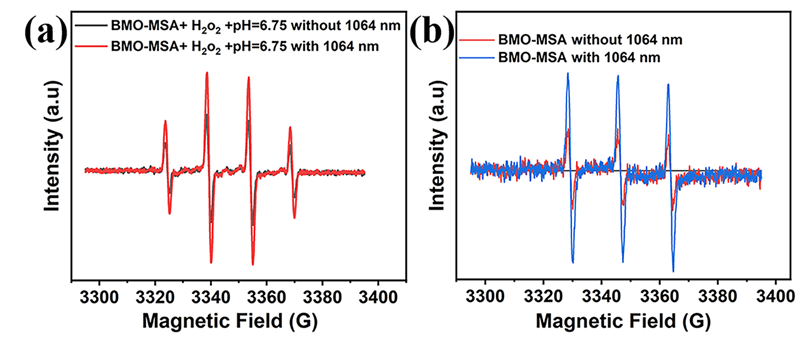
ESR spectrum of (a) ·OH and (b) 1O2 under different conditions. (Image by Shereen)
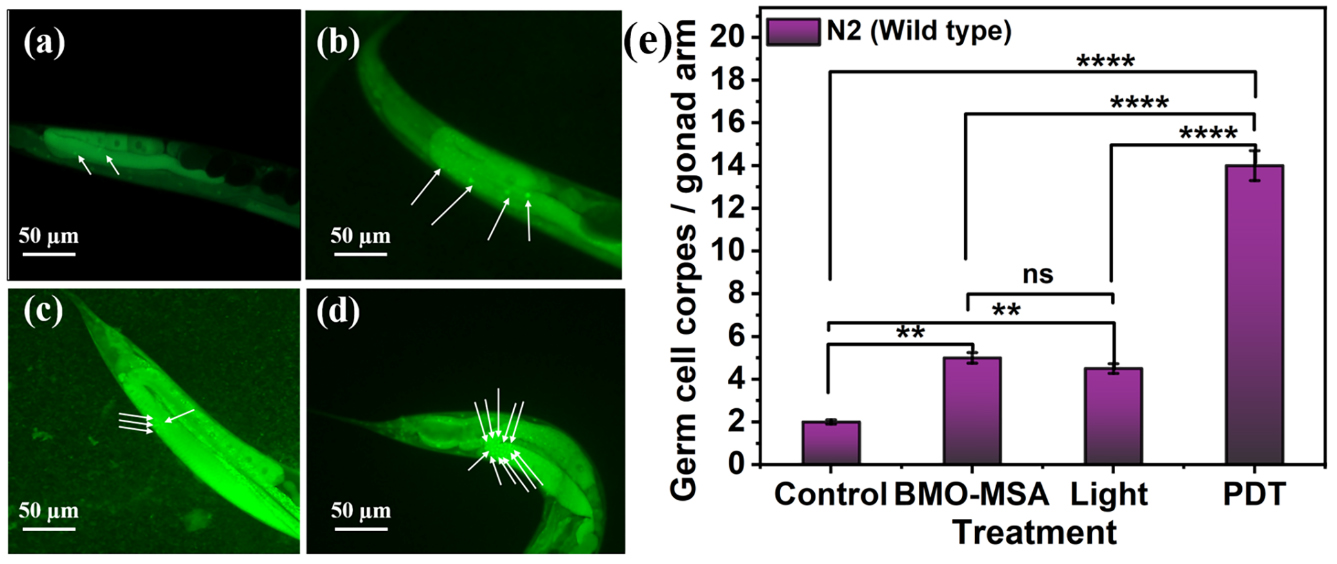
Representative images of germline apoptosis in N2 induced by (a) control, (b) BMO-MSA, (c) light, and (d) PDT. The apoptotic cells are indicated by white arrows. (e) The germ cell corpses in the N2 gonad were induced by BMO-MSA, light, and PDT with control. (Image by Shereen)

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

