
Newsroom
Hydrogen peroxide (H2O2) has a wide range of applications in paper and pulp, textile, wastewater treatment etc. The electrochemical strategy for on-site production of H2O2 via two-electron oxygen reduction reaction (2e- ORR) is a promising alternative. However, the highly sluggish kinetics of 2e- ORR process and the competitive 4e- ORR to H2O severely decrease the yield of H2O2. Therefore, development of electrocatalysts with high activity and selectivity of 2e- ORR is critically important for high-efficiency electrosynthesis of H2O2.
Recently, scientists from the Institute of Solid State Physics (ISSP) of the Hefei Institutes of Physical Science (HFIPS) synthesized a nitrogen-coordinated Co single atom catalyst with superior two-electron oxygen reduction reaction (2e- ORR) performance toward H2O2 production.
In this work, they fabricated a nitrogen-coordinated Co single atom catalyst (Co-SAs/NC) using the coordination regulation strategy. The Co-Nx active sites and nearby oxygen functional groups have a synergetic effect that can promote the H2O2 activity and selectivity.
It's proved that when using Co-SAs/NC as the electrocatalyst, a H2O2 yield of 38.1±1.5 mmol gcat-1 h-1 or (17318±682 mmol gCo-1 h-1) with a faradaic efficiency of ~72.1±4.2% at 0.5 V (vs. RHE) could be achieved in 0.1 M KOH.
These new findings would be very helpful for designing and developing high-efficiency 2e- ORR electrocatalysts for H2O2 production.
This work was financially supported by the Natural Science Foundation of China, the China Postdoctoral Science Foundation, and the CASHIPS Director's Fund.
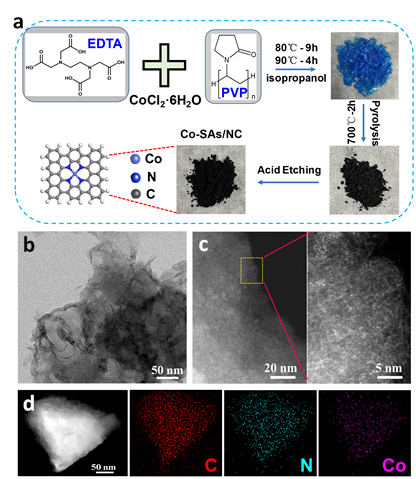
Fig. 1. Schematic illustration of the synthetic process, TEM, aberration-corrected HAADF-STEM and corresponding elemental mapping images of Co-SAs/NC. (Image by HFIPS)
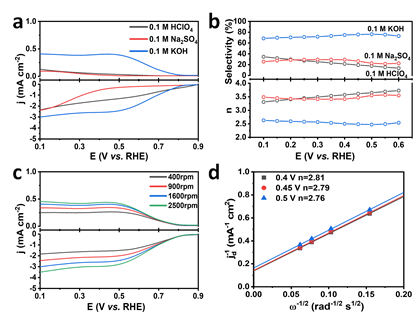
Fig. 2. LSV curves, calculated H2O2 selectivity and electron transfer number (n) of Co-SAs/NC at 1600 rpm in different O2-saturated electrolytes and at different rotation speeds in 0.1 M KOH. (Image by HFIPS)
