
Hydrogen fuel, with the advantages of clean, renewable and of high fuel efficiency, is seen as the "ultimate fuel" and getting more attention around the world. Water electrolysis is an ideal way to produce hydrogen, but it requires active and stable catalysts which make this process more efficient and cheaper.
In a recent study published in Nature Catalysis as the cover story, Prof. WU Yuen's team from the University of Science and Technology of China (USTC) of Chinese Academy of Sciences successfully prepared a kind of Ruthenium (Ru) single atom alloy catalyst, which greatly accelerates the process of water eletrolysis with lower overpotential (220 mV).
By surface defect engineering to capture and stabilize single atoms, this single-atom catalyst is capable of delivering a 90 mV lower overpotential to reach a current density of 10 mA/cm2, and an order of magnitude with longer lifetime than that of commercial RuO2.
Researchers constructed a series of alloy-supported Ru1 in this study using different PtCu alloys through sequential acid etching and electrochemical leaching.
They also found a volcano relation between oxygen evolution reaction (OER) activity and the lattice constant of the PtCu alloys. Through density functional theory investigation, they revealed that the compressive strain of the Pt-skin shell engineers the electronic structure of the Ru1, allowing optimized binding of oxygen species and better resistance to over-oxidation and dissolution.
Compared with Iridium-based systems which have better dissolution resistance, Ru-based ones have more abundant reserves and have been evaluated to be a more active OER catalyst due to its lower overpotential.
This study makes hydrogen production through water electrolysis easier and more efficient, and allows people to see the great potential of hydrogen as an alternative new energy in the future.
Nevertheless, till now, the stability problem of catalyst hasn't been completely resolved, and the reaction system needs further improvement.

Oxygen evolution reaction (OER) and oxygen reduction reaction (ORR) are regarded as two trophies in the field of efficient utilization of hydrogen energy. With Ru-based catalyst, acidic OER process is significantly accelerated. (Image by CUI Jie)
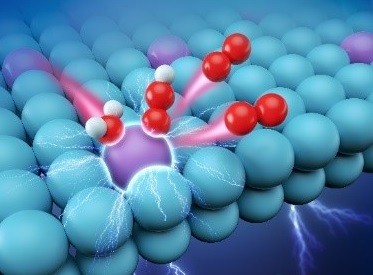
In acidic electrolyte environment, water molecules was adsorbed on the active site Ru atom, and then electrolyzed. Purple, blue, red and white balls represents Ru, Pt, O and H atoms, respectively. (Image by WU Yuen)

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

