The ubiquitin 26S proteasome system is very important for almost all aspects of plant growth and development to degrade misfolded or unnecessary proteins.
Recent studies in human found that in addition to the standard proteasome, there are tissue-specific and stress-specific forms/types proteasome. These specific forms of proteasomes are associated with many diseases, and have been shown to mediate the degradation of particular proteins in specific cellular contexts (e.g. cell type-specific or stress-induced). However, there have been no reports of these cellular context-dependent proteasome forms in plants.
The team led by Prof. XIE Qi at the Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, revealed that as in mammals, plants are able to selectively specify the relative proportions of particular proteasome subunits to form salt stress-specific proteasome forms in Arabidopsis.
26S proteasome consists of two sub-complexes: the catalytic 20S proteasome and the 19S regulatory particle.
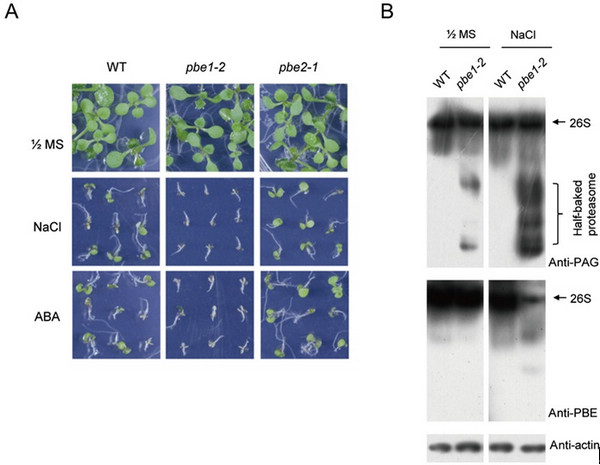
They found that the loss of β5 subunit PBE1 in 20S proteasome function affected the activity and normal assembly of the 26S proteasome in salt-stressed Arabidopsis.
In turn, this proteasome-associated stress modulated the stability of the key transcription factor ABI5 in ABA signaling.
This study is the first report on the specific form of proteasome in plant.
This research is supported by the National Key R&D Program of China and the National Natural Science Foundation of China.
Figure 1. Stress-treated phenotypes of pbe mutants and proteasome assembly affected in PBE1 mutant (pbe1-2) under salt stress. (Image by IGDB)