
Lithium-ion batteries have been applied in portable electronics and electric vehicles due to high energy density, long work life and environmental benignity. But limited lithium sources and high cost seriously have impeded the application of lithium-ion batteries in large-scale energy storage systems.
Because of sodium’s high abundance, low cost, and very suitable redox potential, effort has been made to develop applicable sodium-ion batteries as the substitute of lithium-ion batteries. Recent studies have focused on seeking high performance electrode materials.
The research group led by Prof. YU Yan from University of Science and Technology of China (USTC) of Chinese Academy of Sciences has realized remarkable electrochemical performance of phosphorus-based anode materials for sodium-ion batteries in long cycle life and rate capability by developing one rational strategy. This breakthrough enables sodium-ion battery to replace lithium-ion battery.
Red phosphorus (P) is considered as a promising anode material for sodium-ion batteries, which can deliver a high theoretical sodium-storage capacity of 2595 mAh /g. Prof. YU Yan's group had applied highly order mesoporous carbon to load red P and realized improvement of lithium & sodium storage performance (Nano Lett.). Based on the previous work, researchers devoted to the improvement of sodium storage performance of phosphorus-based materials.
They demonstrated a rational strategy by confining nano-sized amorphous red P into metal-organic framework derived nitrogen-doped microporous carbon matrix, which highly improves the sodium storage performance of phosphorus-based anode materials. When used as anode for sodium-on batteries, the obtained material displays a high reversible specific capacity of ~600 mAh/g at 0.15 A/g and ultralong working life at a high current density (~450 mAh/g at 1 A/g after 1000 cycles).
The prepared red P-based anode material shows great promise for practical application in the next-generation sodium-ion batteries. This design could be extended to prepare other energy storage materials that need efficient ionic and electronic transport.
This work is supported by the National Natural Science Foundation of China, the “Recruitment Program of Global Experts”, the Fundamental Research Funds for the Central Universities, and the Collaborative Innovation Center of Suzhou Nano Science and Technology.
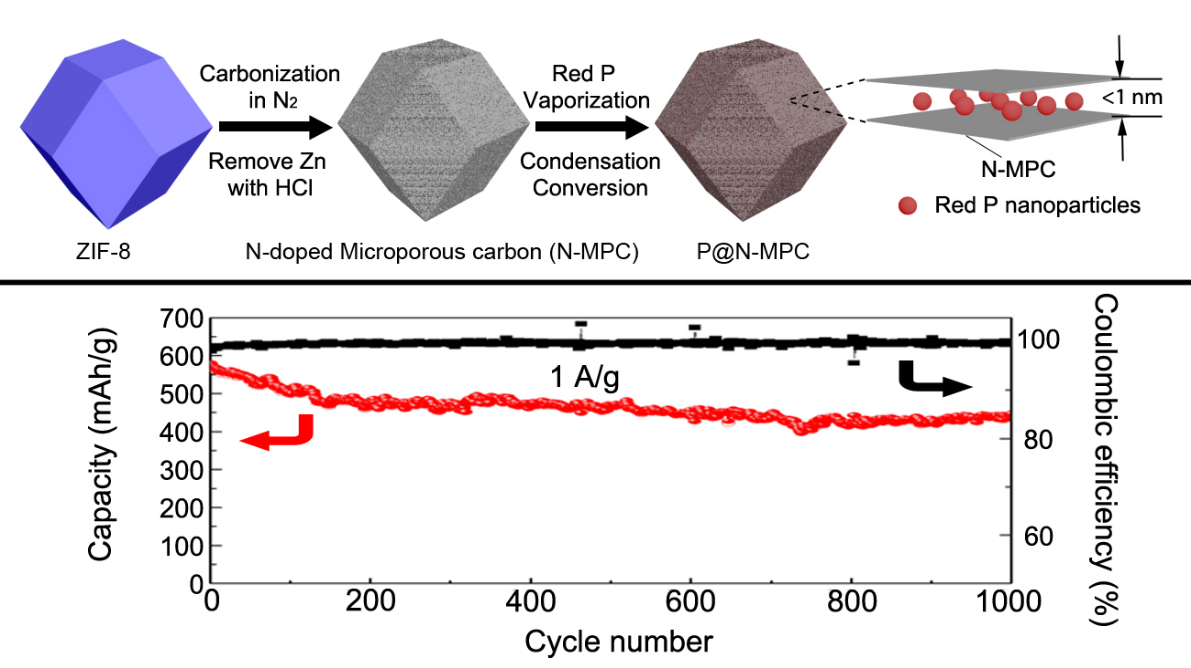
Figure: Schematic illustration and electrochemical performance (Copyright @ John Wiley & Sons, Inc)

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

