
Microglia are the only resident immune cells in the central nervous system (CNS). In the past decade, researchers have revealed that the “resting” (inactivated) microglia in the normal brain are not resting and continuously survey surrounding neural parenchyma through highly dynamic extension and retraction of their processes.
Microglia act not only as immune system guardians of the CNS, but also as versatile sculptors for the normal development and functions of the brain. Deficiency in microglial functions is closely linked to both the generation and development of multiple brain diseases.
Prof. DU Jiulin’s research group at the Institute of Neuroscience, Shanghai Institutes for Biological Sciences of Chinese Academy of Sciences published an invited commentary paper entitled “A Death Trap for Microglia” in Developmental Cell to recommend two research articles published in the same issue of Developmental Cell and the latest issue of Cell Reports respectively on July 26, 2016. These two articles investigated the mechanism underlying the microglial colonization of the brain.
Microglial cells are not born in the CNS, but derive from erythromyeloid precursors (EMPs) present in the yolk sac and immigrate into the CNS from periphery during embryogenesis. How microglia colonize the CNS remained unresolved for a long time due to the technique challenge.
Taking advantage of the non-invasive long-term time-lapse imaging of larval zebrafish, Prof. WEN Zilong’s lab at the Hong Kong University of Science and Technology and Prof. PERI Francesca’s group at the European Molecular Biology laboratory in Heidbelberg Germany published independently two exciting studies.
These studies demonstrateed that neuronal programmed cell death (PCD) during early brain development induces the entry of peripheral microglial precursors into the brain and up-regulating and down-regulating of the level of neuronal apoptosis in the brain could accordingly increase and decrease the number of invading microglia (see Figure). Furthermore, they showed that the lysophosphatidylcholine (LPC), an apoptotic cell-secreted phospholipid which is most likely released by apoptotic neurons, acts as a chemoattractant to attract microglial precursor to immigrate into the brain.
The two studies not only elucidate the mechanism underlying microglial colonization of the brain, which promotes the understanding of early establishment of neuroimmunity, but also reveal the payoff of neuronal PCD, usually considered as an uneconomic physiological process.
Using zebrafish as the animal model, Prof. DU Jiulin’s laboratory has long been engaging in investigating the neural basis for animal behavior and exploring interdiscipline research about neuro-vascular and neuro-immune interactions. As for neuro-immune interaction, the laboratory discovered that the “resting” microglia can dynamically regulate neuronal activity.
This study is recognized as a groundbreaking work to understand the physiological function of microglia and provides new perspective on neural-immune interactions. It has been cited by almost all the related important review papers in the field, and at the end of 2013, it was selected as the most influential works (ranking the first) by Neuron on the journal's 25-year ceremony.
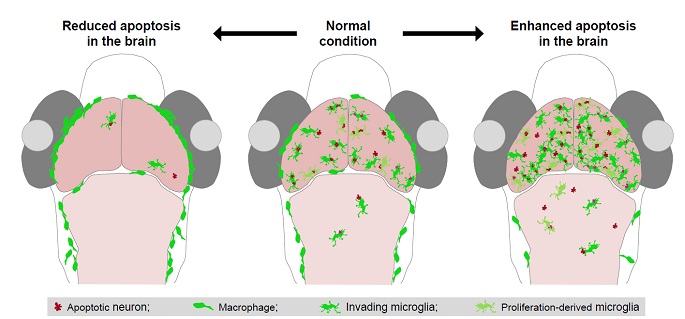
Figure: Developmental Neuronal Apoptosis Induces Microglial Colonization of the Brain. (Image by Dr. DU Jiulin's Lab)

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

