
Newsroom
Surface charges play an important role in the catalytic reaction in photoelectrochemistry. However, the spatial heterogeneities of charge transfer sites and catalytic sites at the electrode/electrolyte interface obscures the surface reaction process.
Recently, a research group led by Prof. LI Can and Prof. FAN Fengtao from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) and their collaborators revealed the linear law between surface charge density and reaction current in photoelectrocatalysis.
This study was published in The Journal of Physical Chemistry Letters on Nov. 2.
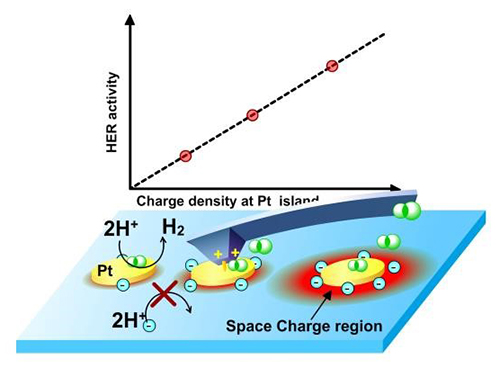
Local HER current on Pt/Ti islands was strictly regulated by the surface charge density in a linear fashion (Imaged by NIE Wei and FAN Fengtao)
The researchers quantified the relationship between the local catalytic current of the hydrogen evolution reaction (HER) and the surface charge density using operando spatially resolved photovoltage microscopy on the Pt/Ti array on the p-Si photoelectrode.
They found that the local HER current could be linearly regulated by the charge density at reactive sites by concurrently adjusting the bias potential and the spacings of the Pt/Ti islands.
"Different from the electrocatalytic process in which the applied voltage changes activation free energy, the uniqueness of applied potential on the photoelectrode was found to determine the charge density of photogenerated minorities and hence the reaction current," said Prof. FAN.
This work was supported by the Fundamental Research Center of Artificial Photosynthesis, the National Natural Science Foundation of China, and CAS Projects for Young Scientists in Basic Research and Interdisciplinary Innovation Team.
