
Newsroom
A research group led by Prof. JIN Shengye from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences revealed an ultrafast and high-yield polaronic exciton dissociation mechanism in two-dimensional (2D) perovskites.
This study was published in Journal of the American Chemical Society on Nov. 3.
2D perovskites are a class of naturally formed quantum well (QW) materials. The exciton binding energy in 2D perovskite is much larger (up to hundreds of meV) than that in their 3D counterparts due to strong quantum and dielectric confinement.
Therefore, it is commonly believed that the photogenerated charge carriers in 2D perovskites are mainly exist in the form of excitons, which are difficult to dissociate into free carriers at room temperature.
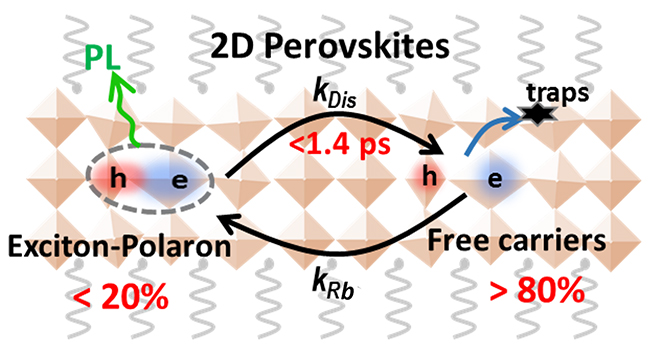
Polaronic exciton dissociation equilibrium in 2D perovskites (Image by SUN Qi, ZHAO Chunyi and TIAN Wenming)
In this study, by using femtosecond photoluminescence up-conversion and transient absorption spectroscopy, the researchers for the first time directly and dynamically observed the ultrafast and highly efficient exciton dissociation in 2D perovskites. This confirmed that free-carriers were the dominant carrier species in 2D perovskites under room temperature, which is conflict with the previous thought.
Moreover, they proposed a polaron-induced exciton dissociation mechanism, where the exciton-polarons were readily formed by strong exciton-phonon coupling as in 3D perovskites, and the polaronic screening effect led to a prominent reduction in binding energy for efficient exciton dissociation.
The researchers demonstrated that the exciton dissociation into free-carriers was a major factor limiting the photoluminescence quantum yield of 2D perovskites by introducing additional nonradiative carrier loss.
"This work reveals a common exciton dissociation property in this class of 2D materials and provides a guideline for the design and rational utilization of 2D perovskites in optical and optoelectronic applications," said Prof. JIN.
The study was supported by the Ministry of Science and Technology of China, the National Natural Science Foundation of China and the Youth Innovation Promotion Association of CAS.
