A joint research team led by Prof. HUANG Jiahui and Prof. QIAO Botao from the
Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS), as well as Prof. SUN Keju from Yanshan University, developed an anti-sintering gold nanocatalyst with high catalytic activity. The results were published in
Nature Communications.
Gold nanocatalysts have exhibited unexpected catalytic activities in many catalytic reactions, and been regarded as one kind of promising catalysts for industrial application. However, their low stability stemmed from the easy sintering of gold nanoparticles is a major barrier.
Strategies such as using the strong interaction between the metal and support, coating the catalysts by inert oxide, utilizing meso-porous materials to confine noble metal particles can effectively improve the sintering resistance of gold nanocatalysts. However, these progresses are achieved at the cost of losing the activity to different extent.
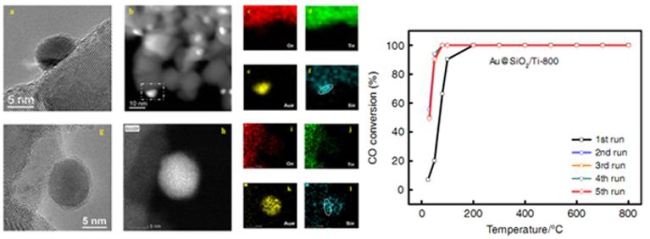
STEM images and EDS mapping of SiO2 modified gold nanocatalyst (left) and CO conversion versus temperature for different cycles (right). (Image by ZHANG Junying)
Recently, the joint research team prepared a SiO2 modified gold nanocatalyst through co-deposition of gold and silica precursors on the TiO2 support and subsequent high temperature calcination.
This method realizes the mixing of gold species and silica species in atomic level. Through the subsequent calcination process, a SiO2 film with only a thickness of a few atom layers was formed, covering the surface of gold nanoparticles.
This catalyst exhibited highly sintering resistant property and gold nanoparticles could maintain at about 6 nm even after 800 °C calcination. It also displayed excellent catalytic property and could realize 100% conversion of CO at 0 °C in CO oxidation.
Experiments together with computational studies revealed that the SiO2 layer over gold nanoparticles not only prevented the growth of gold nanoparticles, but also promoted the adsorption and activation of O2 during CO oxidation, resulting in a high catalytic activity. The finding paves a way for the design and development of gold nanocatalysts with excellent stability and high catalytic activity.


