
Newsroom
Water-induced structural evolution of oxide catalysts is a key issue in heterogeneous catalysis, as it strongly affects the catalysts' active states, stability, and reaction pathways. However, the atomic-scale understanding of how water drives structural reconstruction in oxide catalysts remains unclear.
To address this knowledge gap, a research team led by Profs. FU Qiang and MU Rentao from the Dalian Institute of Chemical Physics of the Chinese Academy of Sciences has uncovered the water-induced dynamic structural evolution of oxides. The study reveals at the atomic scale the dynamic mechanisms of oxidative and reductive hydroxylation of cobalt-based oxide (CoOx) nanostructures in water vapor.
The findings were recently published in National Science Review.
The researchers found that CoOx nanostructures with different initial structures follow distinct hydroxylation pathways in the presence of water. For CoO, water adsorbs dissociatively on the surface, driving its conversion into Co(OH)2 and accompanying the oxidation of Co ions.
In contrast, for CoO2-x surfaces containing both CoO and CoO2 domains, water first reacts with the CoO domains, converting them into Co(OH)2 and forming a Co(OH)2–CoO2-x interface. Subsequent reaction of this interface with water promotes the removal of lattice oxygen, thereby inducing the reduction and hydroxylation of CoO2 domains and ultimately leading to their full conversion into Co(OH)2.
This study clarifies the structural transformation mechanism by which water molecules play both oxidative and reductive roles in different CoOx structures, providing new insights into the role of water in oxide catalysis.
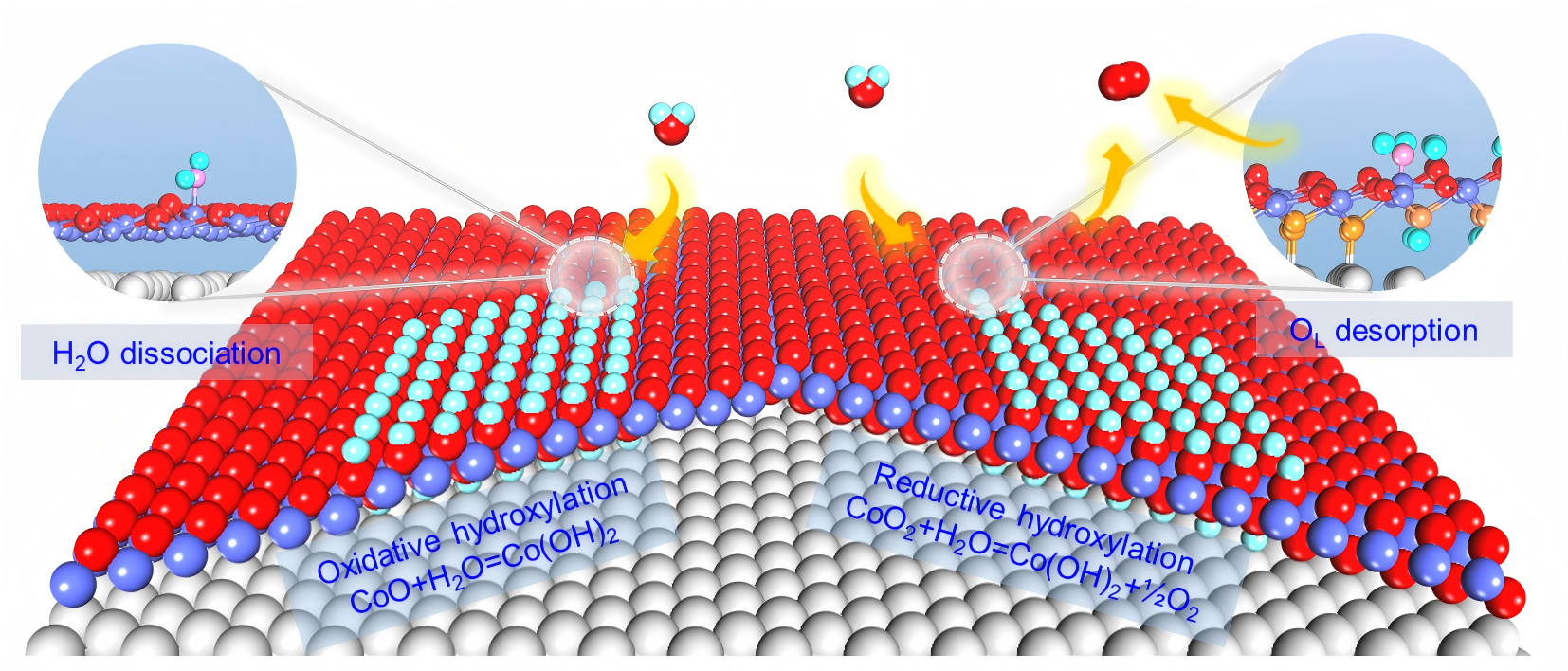
Schematic of reductive and oxidative hydroxylation of CoOx nanostructures in H2O atmosphere. (Image by SUN Xiaoyuan)
