
Newsroom
Neurons have been the main focus in studying Alzheimer's disease, Parkinson's disease, amyotrophic lateral sclerosis, ischemic stroke, and hypoxic-ischemic brain injury. However, increasing evidence suggests that the progression of these disorders is shaped not only by neuronal damage itself, but also the behavior of glial cells, especially microglia and astrocytes.
A study published in Ageing Research Reviews and led by MA Yinzhong's team from the Shenzhen Institute of Advanced Technology (SIAT) of the Chinese Academy of Sciences provided an integrated perspective on how glial cells contribute to disease progression through metabolic reprogramming, inflammatory amplification, and neurovascular dysfunction.
Researchers showed that under physiological conditions, microglia and astrocytes were essential for maintaining central nervous system homeostasis. Microglia continuously surveyed the brain environment and participated in immune regulation and debris clearance, while astrocytes supported neurotransmitter recycling, lipid transport, metabolic coupling, and blood-brain barrier maintenance.
In disease states, however, the researchers showed that both cell types underwent profound functional changes. Microglia shifted from homeostatic surveillance to inflammatory or repair-associated states, while astrocytes adopted reactive phenotypes that may be either neuroprotective or neurotoxic.
Moreover, the researchers found that glial dysfunction was closely linked to metabolism. Activated microglia often switched from oxidative phosphorylation to glycolysis, promoting cytokine release and reactive oxygen species production. Astrocytes, in contrast, often exhibited disrupted lipid metabolism, impaired cholesterol transport, and lipid droplet accumulation, all of which can weaken neuronal support and intensify inflammation.
Furthermore, the researchers found the pathological feedback loops between microglia and astrocytes. Once activated, these cells reinforced one another through inflammatory mediators, complement signaling, metabolites, and extracellular vesicles, creating a self-sustaining cycle of chronic neuroinflammation. This process extended beyond neurons and involved the blood-brain barrier and the neurovascular unit, helping explain why many neurodegenerative and cerebrovascular diseases share common patterns of injury progression.
This study suggests that glial cells should be viewed not simply as inflammatory responders, but as central regulators linking metabolism, immunity, and neurovascular stability. It opens up new avenues for therapeutic strategies aimed at reprogramming glial states and restoring brain homeostasis.
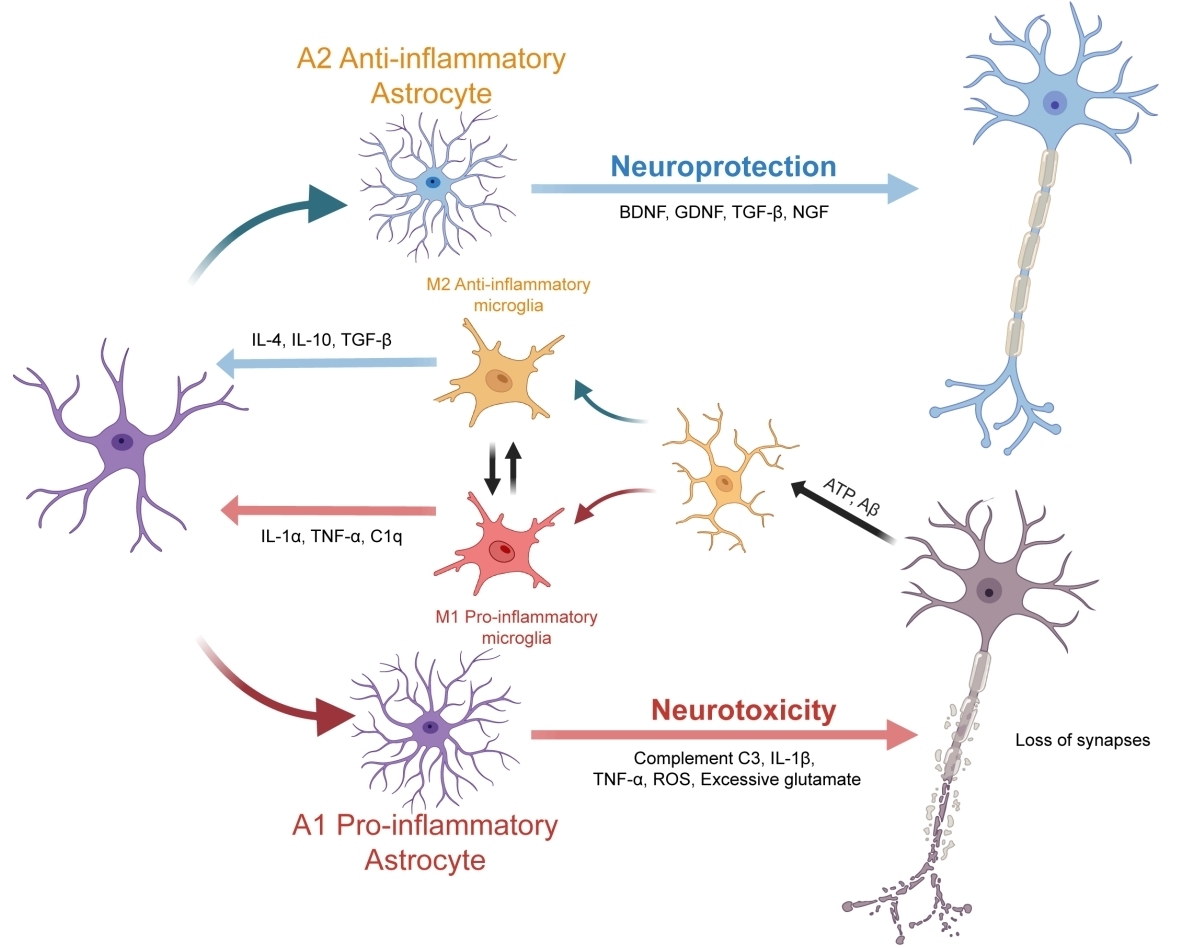
Molecular mechanisms of glial cell activation. (Image by SIAT)
