
Newsroom
The cell's endoplasmic reticulum (ER) plays a central role in protein synthesis, folding, and calcium (Ca²⁺) storage. When damaged, ER-phagy (self-eating) removes affected ER regions via double-membrane vesicles called autophagosomes. However, the mechanisms underlying its initiation, signaling, and membrane origin had previously been unclear.
Now, a research team led by ZHANG Hong from the Institute of Biophysics of the Chinese Academy of Sciences has discovered how mechanosensitive ion channels trigger the selective breakdown of damaged ER fragments via calcium signaling.
The study, published on April 2 in Molecular Cell, provides new insights into ER quality control and suggests potential therapeutic strategies targeting diseases associated with ER dysfunction.
Using advanced techniques including super-resolution multi-SIM live-cell imaging, the researchers observed that under stress conditions, ER sheet subdomains enriched in Ca²⁺ expand and are subsequently engulfed and degraded by autophagosomes.
Electron microscopy and correlative cryo-electron tomography further showed that the membranes of the autophagosomes that enclose these high-Ca²⁺ ER sheets are derived directly from ER remodeling.
The researchers showed that this process depends on the coordinated action of the ER-phagy receptor FAM134B and lipidated LC3.
Further investigation revealed that two mechanosensitive channel proteins, PIEZO1 and TRPV1, are enriched in these high-Ca²⁺ ER sheets. These channels sense changes in calcium levels and trigger localized Ca²⁺ release, generating transient calcium signals.
These Ca²⁺ transients then induce the liquid–liquid phase separation of the autophagy-initiating FIP200 complex, which ultimately initiates ER-phagy.
Disruption of ER calcium homeostasis and defects in ER-phagy have been implicated in a range of diseases, including neurodegenerative disorders and cancer. By revealing how stress-induced, calcium-enriched ER fragments are selectively targeted for degradation, the study offers important clues for understanding the molecular basis of these conditions and highlights potential new targets for therapeutic intervention.
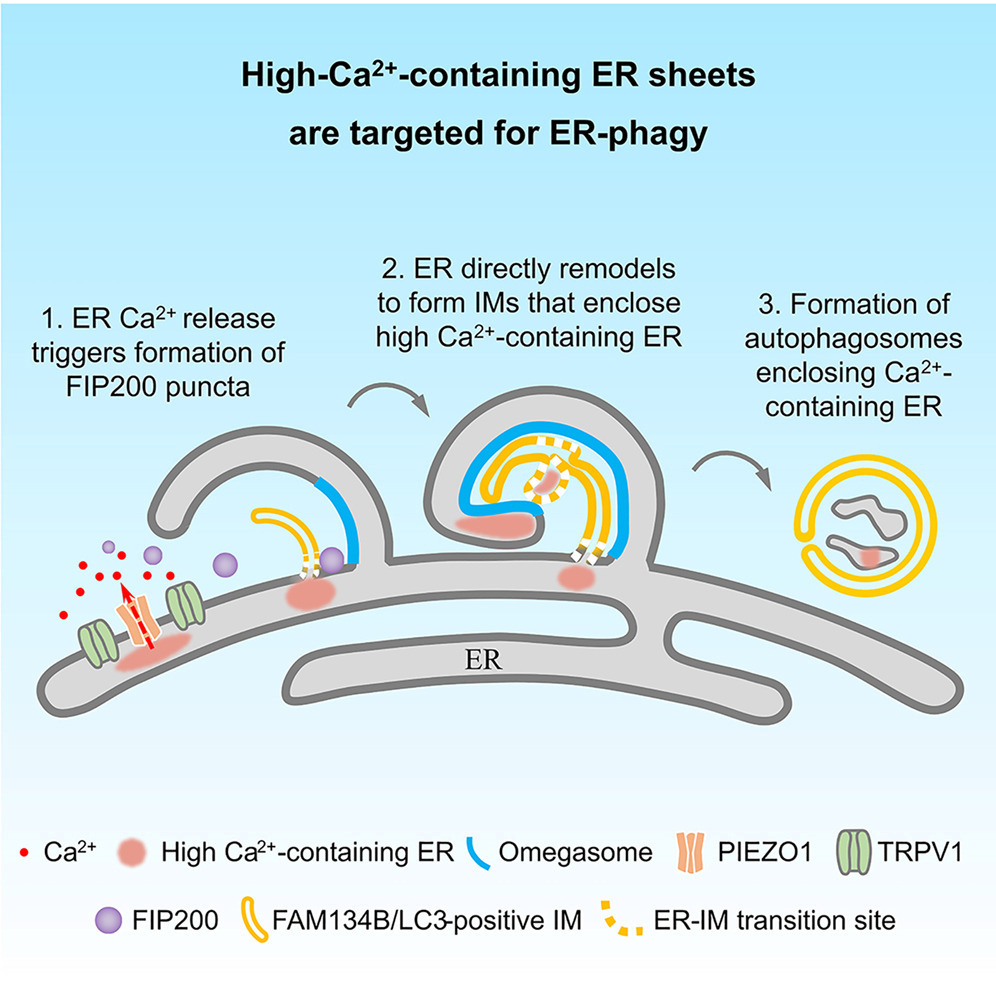
Model of autophagosome formation containing high-Ca²⁺ ER (Image by ZHANG Hong's group)
