
Newsroom
Slow freezing and vitrification cryopreservation are well-established techniques for preserving samples spanning from cells and embryos to simple tissues. For large, complex, and irregular biosamples, vitrification outperforms slow freezing for long-term storage, as it prevents ice crystal formation and mechanical damage through rapid cooling and warming processes.
Optimal vitrification relies on fast, uniform heat transfer across the entire sample. However, during ultra-rapid cooling and rewarming, significant temperature gradients often arise between the interior and surface of an organ. This unsteady heat transfer not only induces uncontrollable ice crystal formation but also triggers massive thermal stress, leading to macroscopic "thermal cracking" of the organ. Traditional heat transfer methods—relying solely on external metal foil wrapping or the perfusion of magnetic nanoparticles—have failed to solve the problem, primarily due to high contact thermal resistance and uneven distribution of internal heat sources.
To address this challenge, a research team from the Technical Institute of Physics and Chemistry (TIPC) of the Chinese Academy of Sciences (CAS) has developed a flexible liquid metal (LM) cryoprotectant composed of eutectic gallium-indium alloys mixed with polyvinylpyrrolidone. Their findings were published in Matter on March 30.
By combining the LM cryoprotectant with an interventional heat transfer (IHT) strategy, the team achieved a material thermal conductivity of 9.3 W/m·K—nearly 10 times higher than that of traditional nanowarming solutions. The LM cryoprotectant adheres snugly and seamlessly to the irregular surfaces of organs, drastically reducing the contact and interfacial thermal resistance that is unavoidable with traditional rigid metal materials.
When perfused into the complex vascular networks of organs, the LM cryoprotectant also exhibits superior electromagnetic thermal properties, enabling uniform volumetric heating. The researchers noted that with gelatin as a transition layer, they can quickly remove and efficiently recycle the high-thermal-conductivity LM cryoprotectant—making the approach biologically safer and more suitable for clinical applications.
Additionally, the team systematically validated the effectiveness of their research across multi-scale biological samples. In vitrification preservation experiments (-196°C) on skin and blood vessels, the post-rewarming viability of skin tissue increased by 1.7 times, while that of vascular tissues rose by 3.6 times. Moreover, the transplanted skin demonstrated enhanced self-healing capabilities.
The study also attempted the cryopreservation of a medium-sized model animal organ with a highly complex structure: the 10mL-scale rabbit kidney. During cooling and rewarming, the maximum surface temperature variation was reduced by 10 to 41.9°C, and thermal stress decreased by two orders of magnitude.
This research establishes a multiscale enhanced heat transfer paradigm based on LM cryoprotectants for conformal cryopreservation, offering transformative potential for large-scale organ banking and transplantation. The IHT paradigm is expected to have broad applications in whole-organ cryopreservation.
This work was supported by the CAS Strategic Priority Research Program, the National Natural Science Foundation of China, and other funding sources.
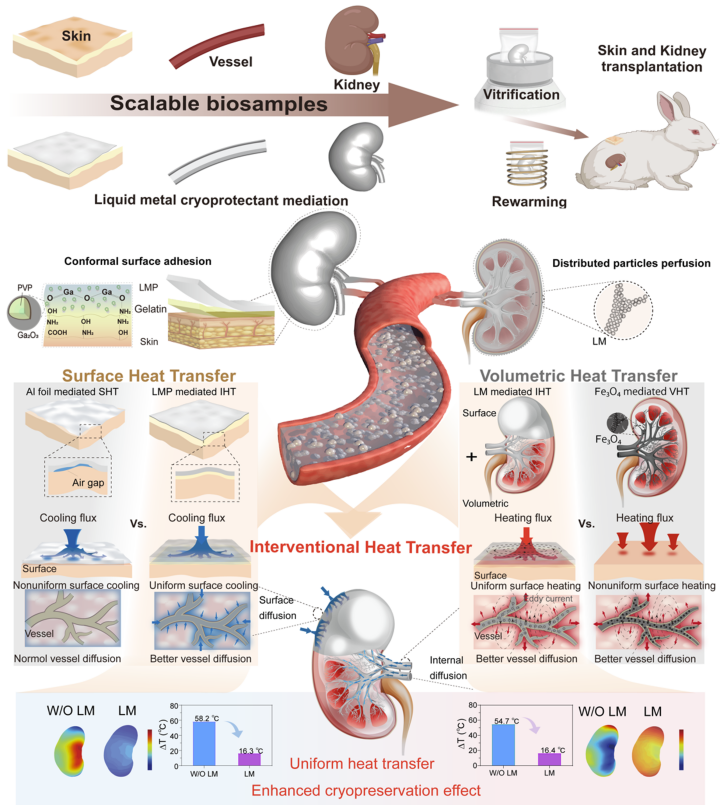
Liquid metal cryoprotectants mediated multi-scale interventional enhanced heat transfer. (Image by TIPC)
