
Newsroom
Researchers from the Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT) of the Chinese Academy of Sciences (CAS) and Hainan University have developed a workflow for detecting metabolites in single cells of corals in situ. This new platform sheds light on the transfer and recycling of carbon and nitrogen across the coral-Symbiodiniaceae-bacteria consortium. It also produces the first single-cell, in-situ metabolic atlas of the coral holobiont.
The study was published in Microbiome on February 27.
To probe thermal adaptation in Symbiodiniaceae, the researchers acclimated a dominant symbiotic clade (C) and a non-symbiotic clade (E) to high temperatures over the long term, generating thermotolerant strains. Using metagenomic profiling, the researchers discovered pronounced metabolic reprogramming. Under heat stress, which typically depletes intracellular proteins, carbohydrates, and lipids, the thermotolerant strains exhibited enhanced protein- and lipid-associated signatures. Some cells accumulated starch, making the strains ideal for phenotypic tracking and targeted screening.
Guided by metagenomic surveys and validated by in vitro assays, the researchers identified a symbiotic bacterium (Muricauda sp.) closely associated with both the coral host and Symbiodiniaceae. Using 13C and 15N substrate labeling in Symbiodiniaceae-bacteria co-cultures, and comparing single-cell Raman spectra before and after feeding, they quantified carbon-nitrogen exchange and uncovered partner-dependent transfer regimes.
In the bacteria-clade E pairing, exchanged metabolites were dominated by amino acid-associated signatures. In contrast, exchange with clade C was more efficient, extending beyond amino acids and carbohydrates to include bidirectional transfer of nucleic acid-associated compounds. This finding implicates exchange directionality and temporal ordering as potential determinants of symbiotic metabolic efficiency.
At the holobiont level, the in-situ workflow tracked biochemical remodeling during bleaching, showing that an increase in bleaching severity coincided with significant depletion of proteins, lipids, and carbohydrates in coral tissues. The researchers then introduced 13C-labeled thermally acclimated Symbiodiniaceae together with 15N-labelled bacteria into bleached coral and monitored recovery alongside metabolic phenotypes.
Although there was little difference in short-term visible recovery among treatments, co-inoculation of clade C with bacteria increased protein-, lipid-, and carbohydrate-associated signals in coral tissues. This is consistent with partial mitigation of physiological impairment and stronger effects than either inoculum alone. Conversely, clade E inoculation resulted in carbohydrate signal accumulation. Microscopy and nucleic acid quantification supported the formation of short-term symbiosis and altered host nutrient partitioning.
"This study provides a new framework for optimizing microbial interactions to help corals adapt to rising temperatures and environmental stresses," said Prof. FU Pengcheng, co-corresponding author of the study. "Our findings pave the way for mitigating coral bleaching and advancing conservation strategies."
"Metaramanomics allows for the real-time monitoring of metabolic exchanges at single-cell resolution, offering new insights into coral symbiosis," added Prof. XU Jian, co-corresponding author from the Single-Cell Center of QIBEBT. "This technique supports the iMAPS (In-situ Metabolic Atlas Projects @ Single-cell) initiative, advancing global research on microbial-host metabolic exchanges and ecosystem conservation."
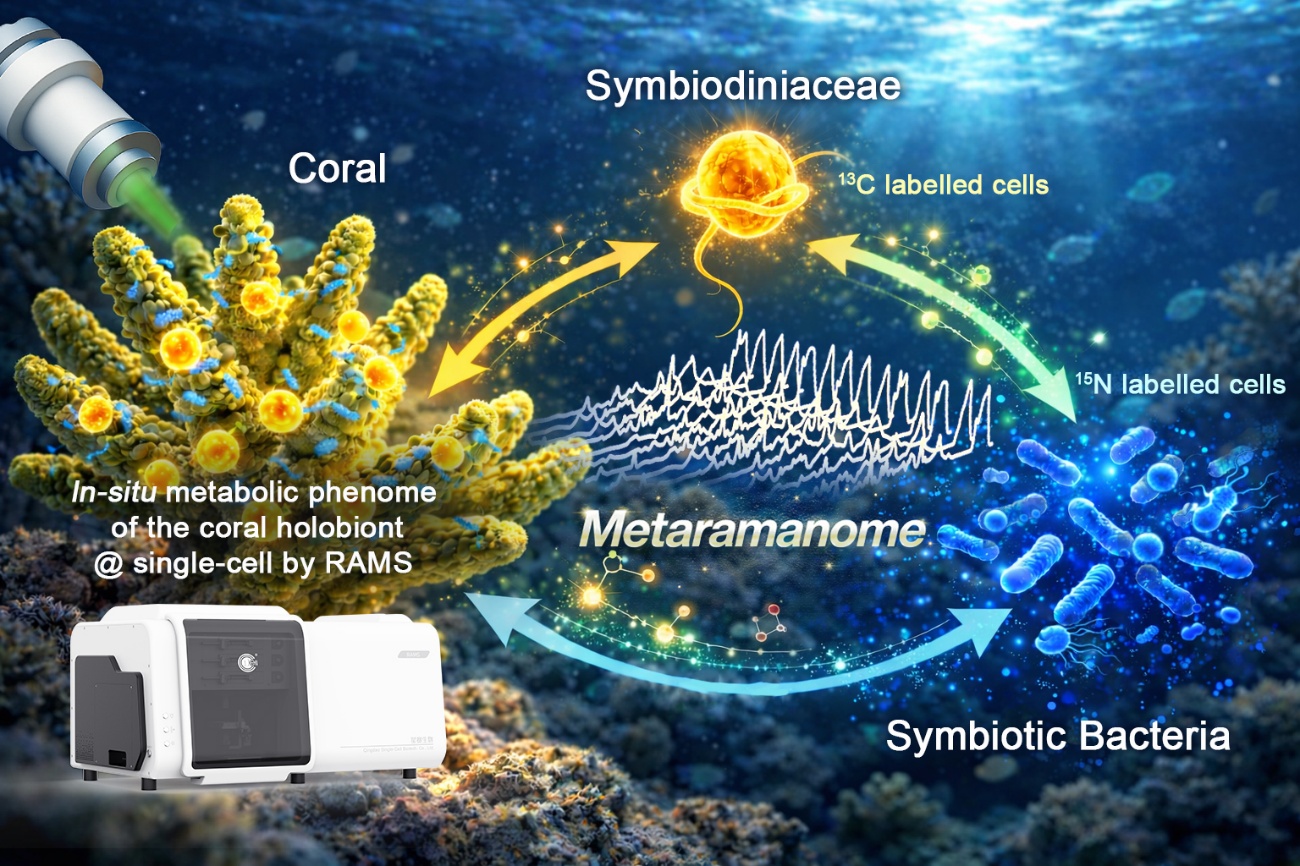
Metaramanome enables a deeper understanding of metabolic interactions within coral-Symbiodiniaceae-bacteria relationships (Image by LIU Yang and SHU Yang)
