
Newsroom
Cilia are hair-like structures found on the surface of many human tissues. They are responsible for driving fluid flow and sensing external signals. Defects in ciliary function can lead to disorders such as respiratory infections, infertility, and hydrocephalus. In a new study published in Structure on March 10, researchers from the Institute of Biophysics of the Chinese Academy of Sciences, and the Center for Excellence in Molecular Cell Science have reported new insights into how the protein Spef1 regulates ciliary beating, providing potential directions for ciliary disease research.
Using structural biology approaches, the researchers first analyzed the architecture of Spef1 and found that the protein contains an intrinsically disordered region and a coiled-coil domain. Unexpectedly, the coiled-coil forms a parallel dimer, rather than the previously speculated antiparallel dimer. This configuration leaves insufficient space for Spef1 molecules to "bundle" central microtubules, suggesting that earlier models could not fully explain how Spef1 regulates the central pair. During these analyses, the researchers observed that Spef1 readily undergoes phase separation, forming droplet-like structures that provided a key clue to its functional mechanism.
Further experiments revealed that Spef1 phase separation depends on both the formation of the parallel dimer and its specific surface charge distribution. Disrupting the dimer completely abolished phase separation, while altering surface charges significantly weakened it. Importantly, when phase separation was impaired, Spef1 lost its ability to bundle central microtubules, indicating that this function relies on phase separation. This mechanism compensates for the limited spatial separation between parallel dimers, providing a new pathway for central microtubule regulation.
The researchers also discovered that Spef1 droplets selectively enrich multiple cilia-related proteins, including tubulin and the motility-associated protein Spag6, both essential for ciliary growth, structure, and function. Strikingly, microtubules grew directly from the tubulin-enriched droplets, suggesting that Spef1 phase-separated condensates recruit and concentrate key proteins at the growing ciliary tip, which is consistent with Spef1's localization at the distal end of developing cilia.
To validate the functional importance of phase separation in vivo, the researchers reintroduced either wild-type or phase separation-deficient Spef1 into Spef1-knockdown ependymal cells. While the mutant protein could still promote central microtubule formation, it failed to restore normal ciliary beating, highlighting a critical link between Spef1 phase separation and ciliary motility regulation.
This study uncovers the molecular mechanism by which Spef1 regulates central microtubule formation through phase separation and directly connects protein phase behavior to ciliary beating. These findings provide new insights into ciliary development and function and identify Spef1 phase separation as a potential target for understanding and treating cilia-related disorders.
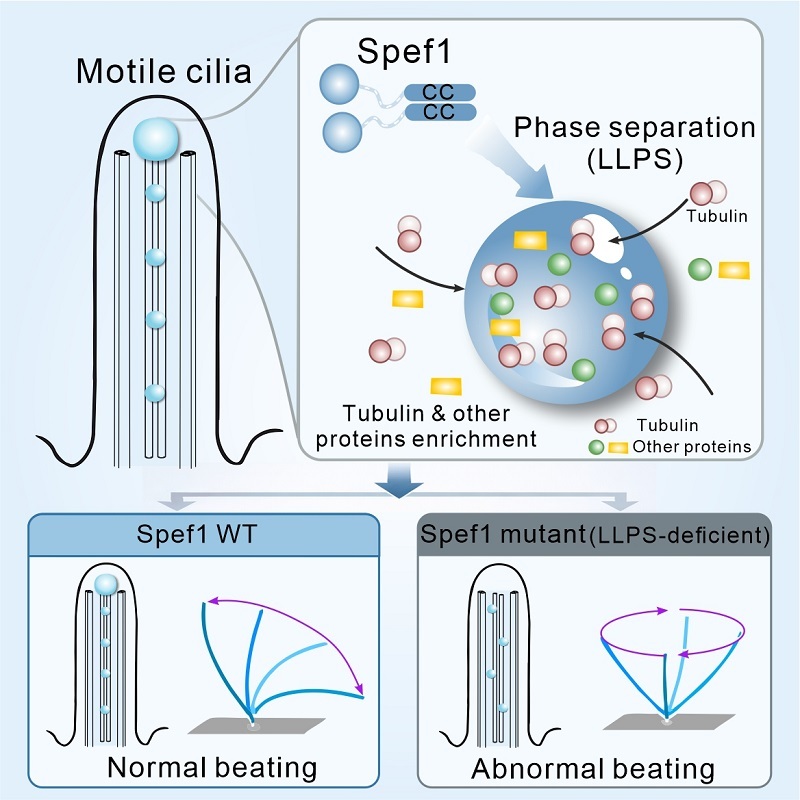
Schematic model illustrating how Spef1 undergoes phase separation to modulate ciliary beating. (Image by FENG Wei's group)
